
Sept. 27-28, 2024 - Symposium for Evolving Therapies and Drug Development in Oncology
Program Description
This two-day conference aimed at improving the knowledge and competence of academic and community oncology providers involved in Early Phase Clinical Trial Development and Management, Phase I/II Clinical Trials Conduct, and Management of Assessments and Safety Parameters involves more than the development of Concepts.
Recent Phase I Studies involving biomarker-guided design in the era of novel therapies has special challenges in selection of studies, patients’ enrollment along with networking with the surrounding institutions to ensure equity and inclusion of patients enrolled in the early phase studies that direct future drug development.
The activity will use a mix of didactic lectures and panel discussions to highlight various aspects of Early Phase Clinical Studies, Precision oncology, and addressing current challenges and opportunities in designing clinical trials in the era of novel therapies.
In-Person Only
This event is being offered as an in-person conference only. There will be no virtual piece or recorded sessions. Attendees will have the opportunity to join the speakers in-person at Inova Center for Personalized Health Conference Center (ICPH) .
Questions & Refunds
If at any time you have questions, please email: [email protected]

Target Audience
- Advanced Practice Providers
- Dietitians
- Family Medicine
- Genetic Counselors
- Internists
- Medical Oncologists
- Nurses
- Nurse Navigators
- Pharmacists
- Physicians
- Radiation Oncologists
- Radiologists
- Residents, Students, and Fellows
- Other Allied Health professionals and Trainees
Learning Objectives
Review challenges and trends in early drug development in the era of novel therapies.
Outline the best practices for the conduct of Phase I/II clinical trials in the era of novel therapies.
Identify phase I clinical trials to match patients’ needs.
Review the challenges and potential solutions for the diverse representation during drug development.
Questions about this event? Contact [email protected]





Parking
Parking is complimentary at the Inova Center for Personalized Health Conference Center (ICPH) . Use surface lots E and F or parking garage for tower C. From the parking garage, take elevator to level 2, walk around the elevators to the bridge over to the conference center.
Travel
Airports and Ground Transportation
Inova Center for Personalized Health Conference Center (ICPH) is centrally located between Washington Dulles Airport (IAD) and Reagan Airports (DCA). Union Station (in Washington, DC) is located about 30-40 minutes from the campus. Both airports along with Union Station offer onsite ground transportation to Fairfax, VA which is about a 30–40-minute drive. Uber or Lyft are also reliable ground transportation options in the DC metro area.
Hotels
Below are the two hotels that are located closest to the Inova Center for Personalized Health Conference Center (ICPH) and offer us discounted rates.
8296 Glass Alley, Fairfax, VA 22031
Phone: 571-327-2277
Inova/corporate rate code: 8296INOVA
3111 Fairview Park Dr, Falls Church, VA 22042
(703) 849-9400
Program Director

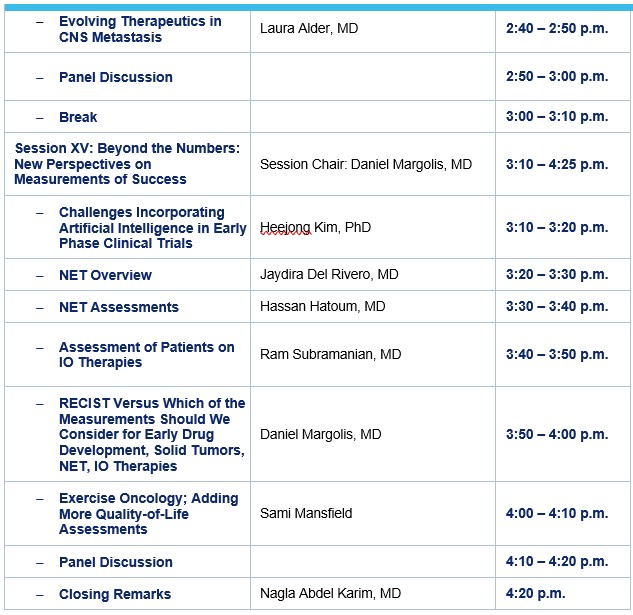
Nagla Abdel Karim, MD
Director of Early Developmental Therapeutics
Inova Schar Cancer Institute
Professor of Medicine
University of Virginia
Advisory Committee:
- Elad Sharon, MD
- Rabih Said, MD, MPH
Drug and Therapeutics Committee:
- Simona Armanca, PharmD
Speaker Disclosure Statement
Inova Health System’s Office of Continuing Medical Education adheres to ACCME’s Standards for Integrity and Independence in Accredited Continuing Education. Any individuals in a position to control the content of a CME activity, including faculty, program directors, and planners are required to disclose all relevant financial relationships with ineligible entities as defined by ACCME. All relevant financial relationships listed for these individuals have been mitigated.
Nagla Karim, MD
Germame Ajebo, MD
Laura Alder, MD
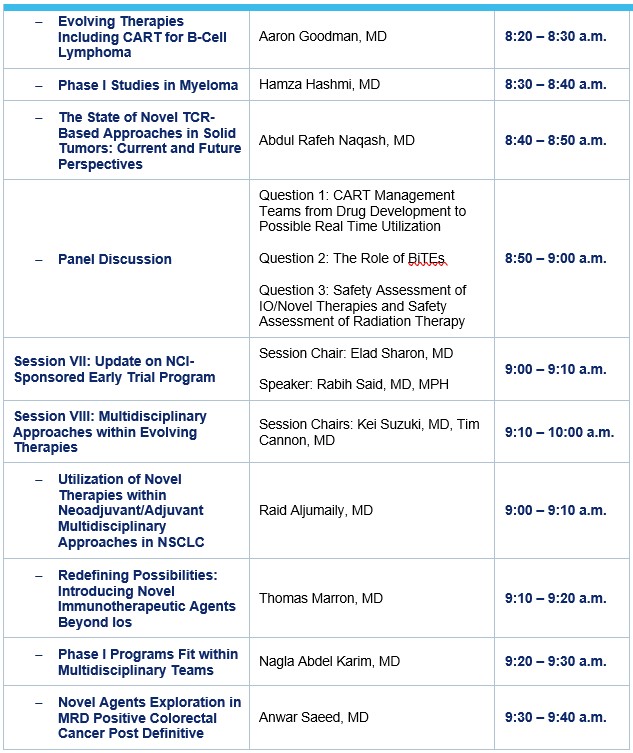
Raid Aljumaily, MD
Simona Armanca, PharmD, BCOP
Minira Aslanova, DO
Nilo Azad
Jennifer Bires, MSW, LCSW, OSW-C
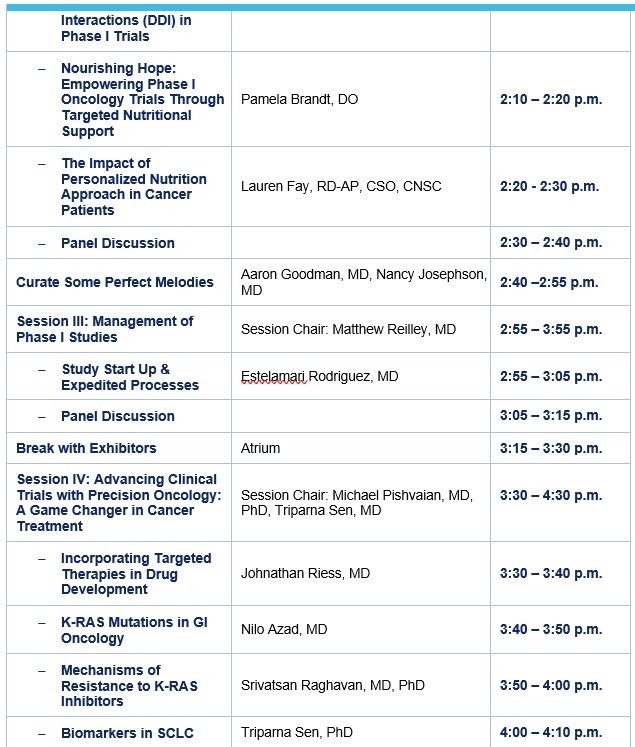
Pamela Brandt, D.O.
Timothy Cannon, MD, Associate Professor at University of Virginia Medical School
Katie Chon, PharmD, RPh
Andrew Ciupek, PhD
Raymund Cuevo, MD
Ayham Deeb, MD
John Deeken, MD
Jaydira Del Rivero
Chris Draft
Bassel El-Rayes
Lauren Fay, MPH, RD-AP, CSO, CNSC
William Figg, Sr.
Stephanie Gaillard, MD PhD
Joseph Gibbons, M.D>
Aaron Goodman, MD
Krishna Gunturu, Medical Oncologist, MD
Hamza Hashmi, M.D.
Yuxia Jia, MD, PhD
Christopher Johnson, MD, PhD
Nagla Karim, MD
Maya Khalil, MD
Chul Kim, MD
Heejong Kim
So Yeon Kim, MD
Chung-Han Lee, MD/PhD
Sami Mansfield, BA, ACSM-CET, PN-1
Daniel Margolis, MD
Thomas Marron
Stephen Medlin, DO
Dalia Mobarek, MD
John Morris
Abdul Rafeh Naqash, MD
Nina Neill, MD, MS
Ishan Patel, MD
Michael (Mike) Pishvaian
Faran Polani, MD
Srivatsan Raghavan, Srivatsan Raghavan, M.D., Ph.D.
Matt Reilley, MD
Jonathan Riess, MD, MS
Estelamari Rodriguez
Radha Rohatgi, MD
Anwaar Saeed, MD
Rabih Said, MD, MPH
Bobby Se, MD
Triparna Sen, PhD
Elad Sharon, MD
Noha Soror, MD, MSc
Janakiraman Subramanian, MD, MPH
Kei Suzuki
Brian Van Tine, MD, PhD
Raymond Wadlow, MD

Inova CME Accreditation
The Medical Society of Virginia is a member of the Southern States CME Collaborative, an ACCME Recognized Accreditor. Inova Health System Office of Continuing Medical Education is accredited by the Southern States CME Collaborative to provide continuing medical education for physicians.
Credit Designation
The Inova Office of Continuing Medical Education designates this live educational activity for a maximum of 11.25 AMA PRA Category 1 Credit(s) ™. Physicians should only claim credit commensurate with the extent of their participation in the activity. Physicians may claim up to 11.25 credits in Type 1 CME on the Virginia Board of Medicine Continued Competency and Assessment Form required for renewal of an active medical license in Virginia.

Pharmacy Credit
VSHP accredited by the Accreditation Council for Pharmacy Education (ACPE) as a provider of continuing pharmacy education. Participants of the session who complete the evaluation and provide accurate NABP e-Profile information will have their credit for 5.25 contact hours (0.525 CEU) submitted to CPE Monitor as early as 1 day after the event and no later than 60 days after the event. Please know that if accurate e-Profile information is not provided within 60 days of the event, credit cannot be claimed after that time. The participant is accountable for verifying the accurate posting of CE credit to their CPE Monitor account within 60 days.
UAN #0108-9999-24-083-L01-P
Link to Claim Pharma Credits Opens, Saturday, Sept. 28 at 12:30pm ET: https://www.lecturepanda.com/a/24september28-inova

ABIM MOC
Successful completion of this CME activity, which includes participation in the evaluation component, enables the participant to earn up to 3 MOC points in the American Board of Internal Medicine’s (ABIM) Maintenance of Certification (MOC) program. It is the CME activity provider’s responsibility to submit participant completion information to ACCME for the purpose of granting ABIM MOC credit.
Nurse Credentialing FAQs
For the purpose of recertification, the American Nurses Credentialing Center (ANCC) accepts AMA PRA Category 1 Credits™ issued by organizations accredited by the ACCME. For more information visit, https://www.nursingworld.org/certification/faqs/
Available Credit
- 3.00 ABIM MOC
- 11.25 AMA PRA Category 1 Credit™
Thank you Exhibitors! Our event would not be possible without the generous support from our sponsors.
- Abbvie Oncology
- Accord BioPharma
- Agios
- Alexion Pharmaceuticals
- Amgen
- AstraZeneca Oncology
- Boehringer Ingelheim - Medical
- Bristol-Meyers Squibb
- Daiichi Sankyo
- Eisai
- EMD Serono
- Exelixis
- Genentech, Inc.
- Gilead
- Incyte
- Jazz Pharmaceuticals, Inc.
- Lantheus
- Lilly - A Medicine Company
- Merck
- Mirati Therapeutics
- Natera
- OncoHost
- Pfizer Oncology, Lung Cancer
- Pfizer, Inc.
- Pharmacosmos Therapeutics, Inc.
- Regeneron
- Servier Pharmaceuticals
- Taiho Oncology, Inc.
- Takeda Oncology
- Tempus AI
- Yunu
Interested in Exhibiting?
To confirm your space as an exhibitor, complete the exhibitor registration form and return to [email protected].
General Exhibit Booth: $3,000
- 6' undraped table with two chairs
- Conference registration for two representatives
- Verbal recognition the day of the conference with signage, podium mention
- and on screen during the conference
- Company name on:
- Exhibitor page on event website
- Marketing emails
- Introduction slides, break slides, and closing slides
- Post-event follow-up attendee email
Premium Exhibitor Booth: $4,500
- 6' undraped table with two chairs
- Conference registration for three representatives
- Access to sit-in on educational sessions
- Verbal recognition the day of the conference with signage, podium mention
- and on screen during the conference
- Company name on:
- Exhibitor page on event website
- Marketing emails
- Introduction slides, break slides, and closing slides
- Post-event follow-up attendee email
- Attendee registration list
Nonprofit Organizations Booth - $500
(501c3 must be submitted for approval)
- 6' undraped table with two chairs
- Conference registration for two representatives
- Verbal recognition the day of the conference with signage, podium mention
- and on screen during the conference
- Company name on:
- Exhibitor page on event website
- Marketing emails
- Introduction slides, break slides, and closing slides
- Post-event follow-up attendee email
Download the full prospectus here! ![]() <span style="color:#3498db">Evolving Therapies September 2024 PROSPECTUS FORM.pdf</span>
<span style="color:#3498db">Evolving Therapies September 2024 PROSPECTUS FORM.pdf</span>
Questions? Email Christina Fontana for more information.

 Facebook
Facebook X
X LinkedIn
LinkedIn Forward
Forward